Case Study
Patient A,
Male, American
Table of Contents
Patient Overview
Age at time of treatment: 18 – 25
Injury Level: C7
Treatment Received: Stem Cells, Epidural Stimulation
Location of Treatment: Thailand
Time between injury and treatment: —
Date of Surgery: 02/09/2018
Date of Discharge: 05/10/2018
Condition on Admission
Patient sustained a C7 fracture-dislocation on November 2, 2016. He experienced subsequent spinal cord myelomalacia (softening of the spinal cord) and has minimal motor or sensory function below the injury level. He has neurogenic bowel and bladder. Patient does not suffer severe spasticity but does suffer moderate to severe neuropathic pain, particularly in the upper extremities. This is managed by high doses of Gabapentin (600 mg, 4 times a day). Patient is able to move his upper limbs and is independent in his daily activities.
Previous Therapies & Treatments
Prior to coming to Verita Neuro, patient received stem cell treatment in 2017, as well as physical and occupational therapy.
Patient sustained a C7 fracture-dislocation on November 2, 2016. He experienced subsequent spinal cord myelomalacia (softening of the spinal cord) and has minimal motor or sensory function below the injury level. He has neurogenic bowel and bladder. Patient does not suffer severe spasticity but does suffer moderate to severe neuropathic pain, particularly in the upper extremities. This is managed by high doses of Gabapentin (600 mg, 4 times a day). Patient is able to move his upper limbs and is independent in his daily activities.
Verita Neuro Treatment Received
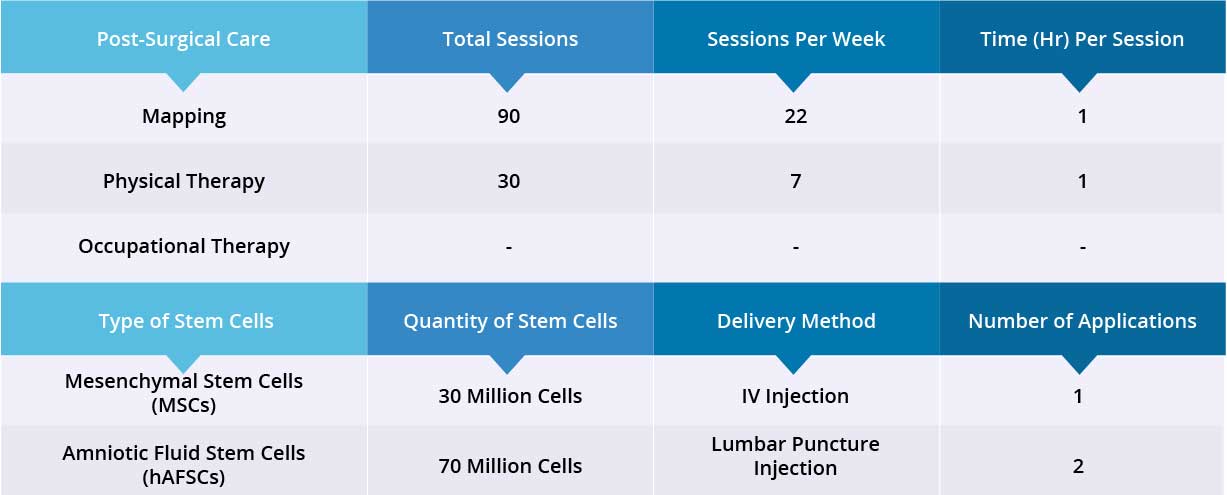
After a spinal MRI scan and comprehensive blood work, patient underwent laminectomy and implantation of the epidural stimulation device. The device is the ‘Medtronic Restore Advance 16-electrode MRI Compatible Device’. The surgery was completed without significant adverse effects and the surgical wound healed normally. No serious complications were reported during the hospital stay. Patient also received 100 million Amniotic Fluid Stem Cells (hAFSC) through a combination of IV injection and lumbar puncture injection. Device Mapping and physical therapy were carried out after surgery for 35 days, and the patient was discharged.
Results
- Motor Functions
- Sensory Functions
- Autonomic Functions
Patient’s gross motor skills have improved significantly when Epidural Stimulation device is switched on, including ankle, hip and knee flexion, and knee extension (kicking out). Patient has good static sitting balance and no support is needed. However, during dynamic sitting, balance is poor due to weak lower trunk muscles. Static standing balance is good at the parallel bar.
There was no noticeable change in bladder and bowel functions. Patient is still taking the same dose of Gabapentin (600 mg, 4 times a day), therefore no change was visible in neuropathic pain. After 35 days, patient was discharged and will continue his physiotherapy back home.
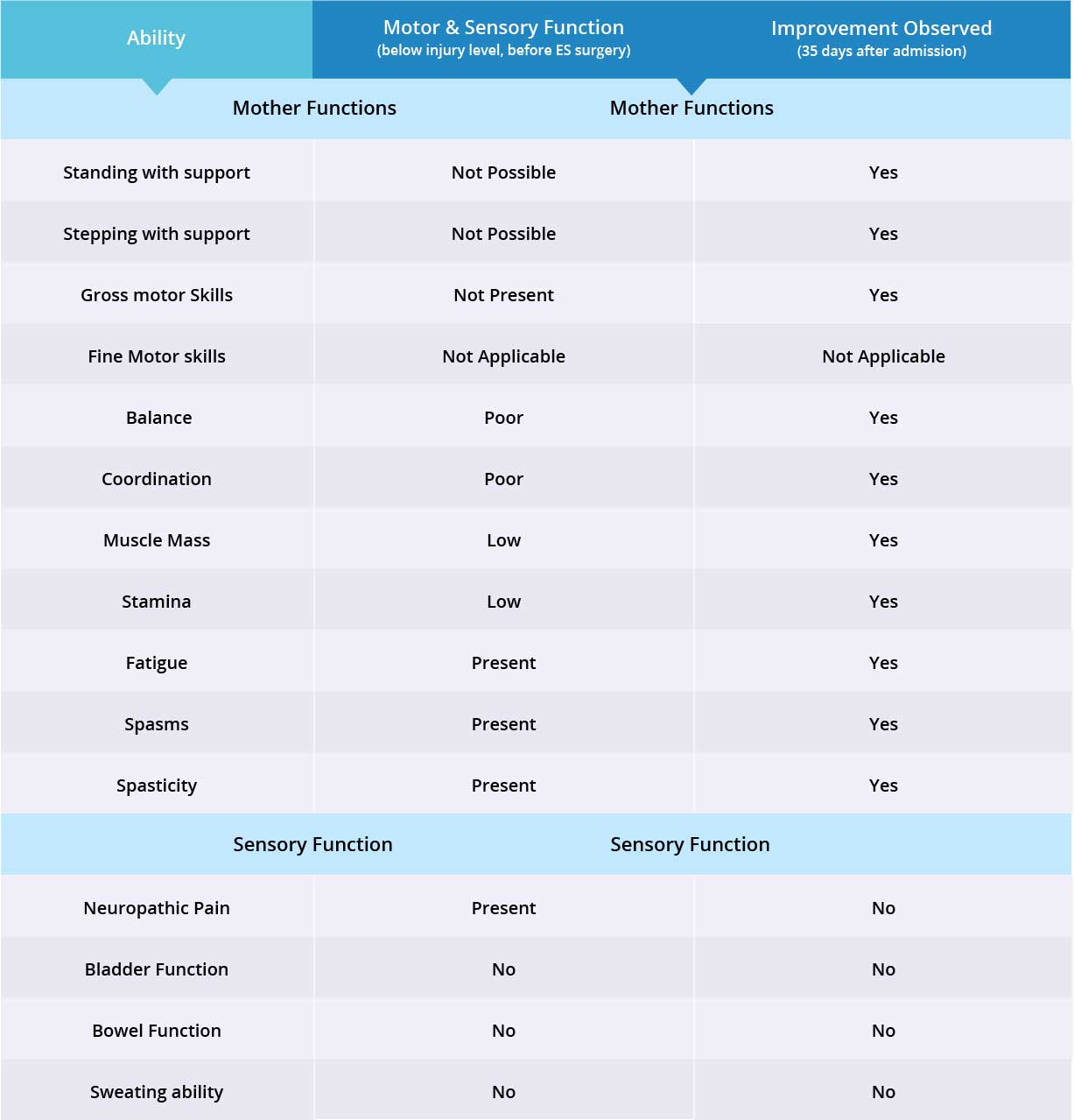
Improvements are monitored in 15 targeted areas: 11 Motor areas and 4 Sensory areas. However, the number of targeted areas may vary depending on patient’s condition prior to admission. If patient does not experience symptoms in certain Motor/Sensory functions, or is not impaired in a specific targeted area prior to surgery, it is excluded from the report (Not Applicable). If there is progress in any given area — either mild, moderate, or significant — it is measured and reported as positive (“Yes”). No improvement, the existence of pain or spasms, or an inability to perform a measured function is reported as “No”.
Results Interpretation
In this patient, fine motor skills were normal, therefore 14 areas instead of 15 were targeted. Motor function improved in 10 out of 10 targeted areas when the Epidural Stimulation device was switched on. There was no change in neuropathic pain. Patient has not noticed any changes in Sensory Function areas, and more feedback will be collected after 3 months to note any improvements made by regenerative medicine treatment. Overall, improvements were recorded in 10 out of 14 targeted Motor and Sensory Function areas.
Three Month Follow Up
- Motor Functions
- Sensory Functions
- Autonomic Functions
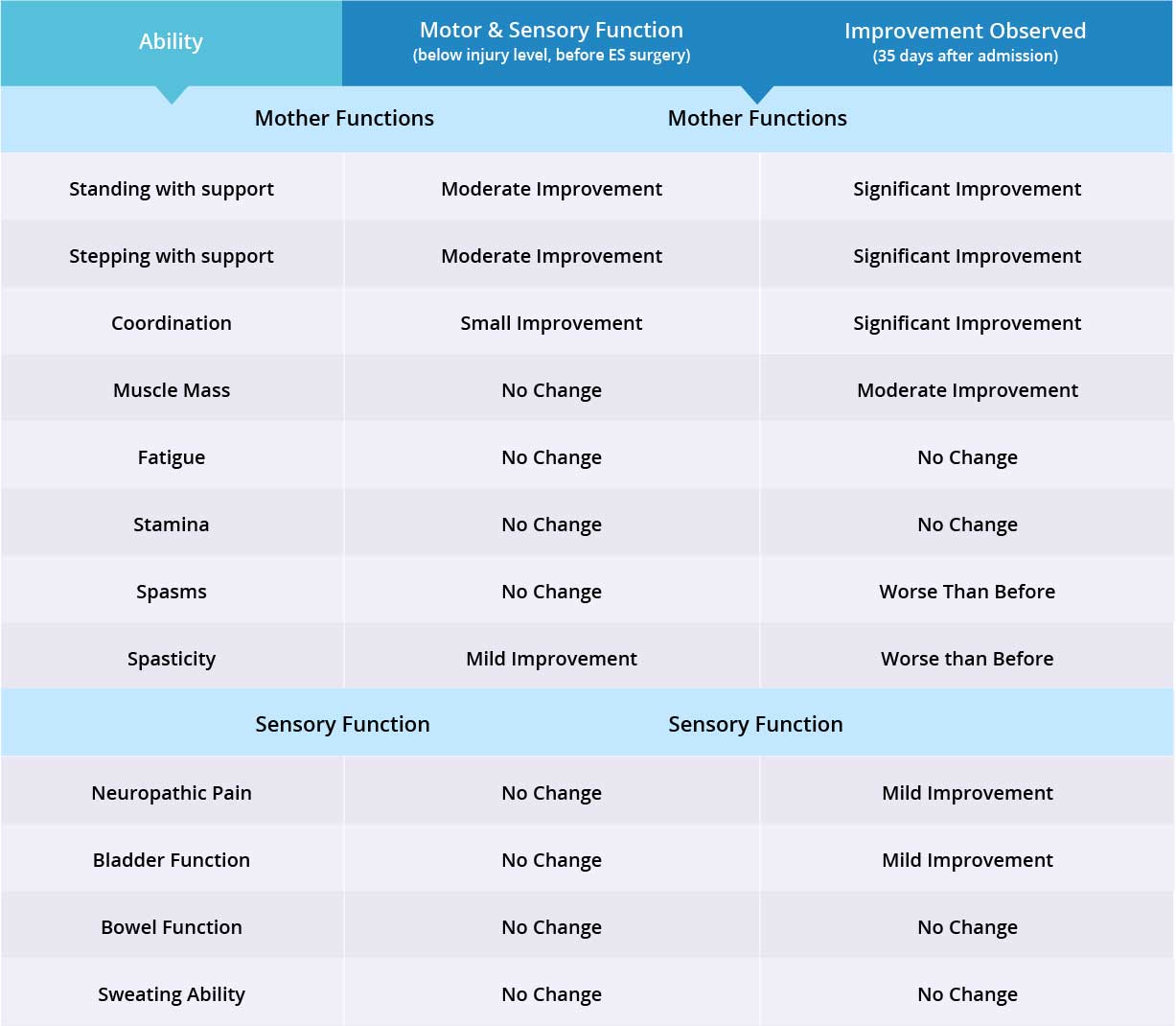
Overall, the patient is very satisfied with his outcomes after the Epidural Stimulation surgery. He will continue his physical therapy and another follow-up call will be made in three months time.
Six-Month Follow Up
- Motor Functions
- Sensory Functions
- Autonomic Functions
The patient has noticed significant improvements during standing and stepping exercises. The patient is still able to stand up straight, has good static standing balance, and improved trunk control. Patient is still able to lock his hips and bears weight equally on both legs. However, patient is still not able to lock his knees completely and requires assistance in keeping his knees locked.
During stepping exercises, the patient is still able to lift his feet and set them down by himself without any assistance. Significant improvement was noticed in the patient’s foot coordination while taking steps. Patient is able to lock his left knee the majority of the time, however, requires assistance in locking his right knee. Patient also reports improvement in strength and stamina, his core muscles have become stronger.

